FDA Launches One-Day Inspectional Assessments Pilot
The FDA has launched a pilot program introducing one-day inspectional assessments across medical products, biologics, clinical research, and food sectors to support more targeted and efficient oversight.
FDA Updates Coding Resources for Medical Device Reports: Key Changes from IMDRF 2025 Maintenance
FDA updates MDR adverse event codes under IMDRF 2025. Key changes may impact manufacturer reporting, vigilance systems, and data consistency.
FDA Issues Draft Guidance on Compliance Policy for Certain NIOSH-Approved Respirators
FDA releases draft guidance proposing reduced regulatory requirements for certain NIOSH-approved respirators. Understand the impact on medical device manufacturers.
FDA launches consultation on AI use in early-phase clinical trials
FDA opens consultation on a pilot program to assess artificial intelligence in early-phase clinical trials, focusing on efficiency, safety, and decision-making.
FDA Issues Draft Guidance on NGS-Based Safety Assessment for Genome Editing Therapies
FDA releases draft guidance on next-generation sequencing (NGS) for genome editing safety assessment. Key implications for gene therapy manufacturers and IND submissions.
FDA Updates eMDR Framework with ESG NextGen Submission Portal
The FDA has replaced the WebTrader system with the ESG NextGen portal for eMDR submissions. Learn what this means for medical device manufacturers and reporting obligations under 21 CFR Part 803.
FDA Issues New Guidance on Bioanalytical Method Validation for Biomarkers
FDA issues new guidance on biomarker validation. Learn key requirements and what manufacturers must consider for global regulatory compliance.
FDA Updates SOPP 8001.4 on Proprietary Name Review for Biologics
FDA releases SOPP 8001.4 v9 on proprietary name review for biologics. Key implications for manufacturers and global regulatory strategy.
FDA Updates SOPP 8212 on Breakthrough Therapy Designation for Biologics
FDA updates SOPP 8212 on breakthrough therapy designation. Key changes in timelines, communication, and lifecycle management for manufacturers.
FDA launches READI-Home Innovation Challenge focused on reducing hospital readmissions
The FDA launches the READI-Home Innovation Challenge to support medical devices aimed at reducing hospital readmissions through home-based care solutions.
FDA expands TAP Pilot: increased scope and participation
FDA expands its TAP Pilot with more devices and new areas. Key updates and what medical device manufacturers need to know.
FDA updates SOPP 8412 on product labeling review
FDA releases SOPP 8412 Version 10 detailing labeling review procedures for drugs and biologics under BLA, NDA, and ANDA pathways.
FDA Publishes Updated Guidance on Pyrogen and Endotoxins Testing
FDA releases updated guidance on pyrogen and endotoxin testing. Key clarifications for medical device and pharma manufacturers.
FDA Issues Final Guidance on Medical Devices with Indications Associated with Weight Loss
The FDA has issued final guidance on medical devices with indications associated with weight loss, outlining regulatory expectations for clinical studies, non-clinical testing, and benefit-risk evaluation.
FDA Updates Recognized Biocompatibility Standards – Recognition List No. 065
FDA Recognition List 065 updates biocompatibility standards, including ISO 10993-4 Amendment 1 and multiple ASTM revisions. Impact for manufacturers.
FDA Publishes Draft ICH E22 Guideline on Patient Preference Studies for Public Consultation
The FDA has published a draft guidance adopting the ICH E22 guideline on Patient Preference Studies. Learn what this means for manufacturers involved in global development programmes.
FDA Issues Updated Final Guidance on Cybersecurity in Medical Devices
FDA issues final guidance on cybersecurity in medical devices, outlining QMS, premarket documentation, and Section 524B requirements for manufacturers.
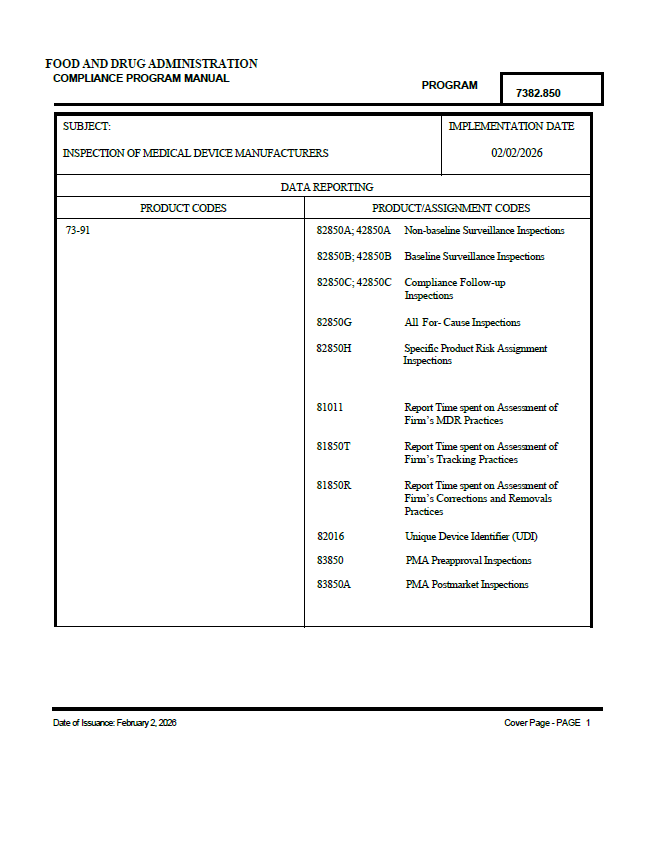
FDA Implements Updated Medical Device Inspection Compliance Program
FDA implements Compliance Program 7382.850, aligning medical device inspections with the Quality Management System Regulation and ISO 13485.
FDA Publishes Updated Guidance on Clinical Decision Support Software
The FDA has issued updated guidance on Clinical Decision Support (CDS) software, clarifying which software functions are excluded from the medical device definition under U.S. law.
New FDA Draft Guidance Sets Bar for Cuffless Blood Pressure Devices – Clinical Performance Now in Focus
The FDA has released a draft guidance outlining new clinical performance requirements for cuffless non-invasive blood pressure devices. Manufacturers using wearable sensors or AI must now meet specific testing standards, including ISO 81060-2. Learn how these updates affect premarket submissions and regulatory strategies.