Swissmedic Updates “Formal Requirements” Guidance – Version 20.0 Now in Force
Swissmedic has released Version 20.0 of its “Formal Requirements” guidance, effective 1 March 2026. Learn what pharmaceutical manufacturers need to know about submission formats, documentation requirements and parallel import updates.
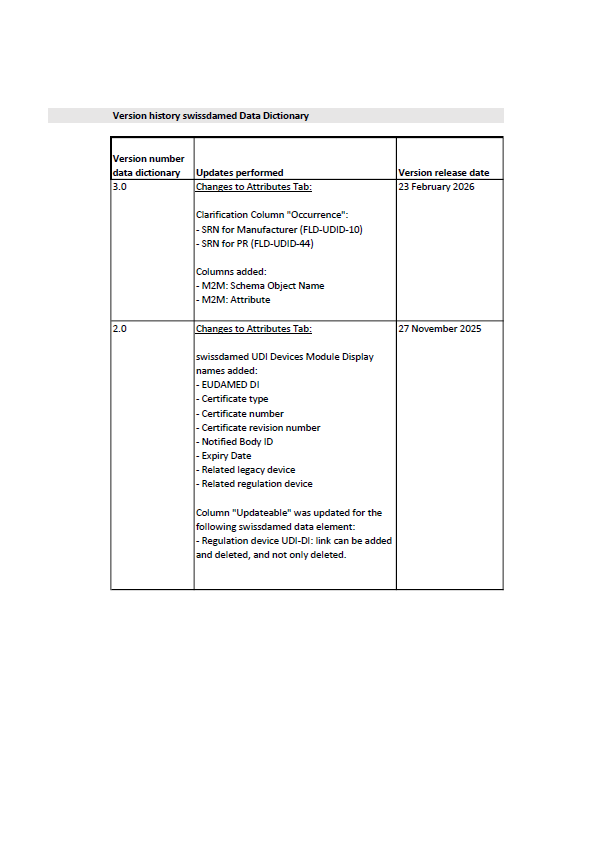
Swissmedic Updates swissdamed UDI Data Dictionary
Swissmedic has published version 3.0 of the swissdamed UDI Data Dictionary, introducing clarifications to SRN fields and new machine-to-machine attributes for improved data mapping.
Team-NB Raises Concerns Over Proposed MDR and IVDR Revision
Team-NB warns that proposed MDR and IVDR revisions could reduce regulatory scrutiny and create unintended consequences for EU manufacturers, despite improvements in digitalisation and innovation pathways.
TGA Publishes Updated Guidance on the Regulation of Software-Based Medical Devices – Key Considerations for Manufacturers
TGA publishes updated guidance on software-based medical devices. Key regulatory implications for manufacturers supplying SaMD in Australia.
IMDRF Publishes Final Guidance on the Selection of Adverse Event Terminology
IMDRF publishes final guidance on the selection and consistent use of adverse event terminology (AET) codes for medical devices and IVDs.
IMDRF Publishes 2026 Playbook for Medical Device Regulatory Reliance Programs
IMDRF releases its 2026 Playbook for Medical Device Regulatory Reliance Programs, outlining work-sharing, recognition and abridged pathways.
TGA Updates PRAC Guidance: What Sponsors and Manufacturers Need to Know in 2026
The TGA updated its PRAC recall procedure in February 2026. Here’s what medical device and medicine manufacturers must know about compliance and market actions in Australia.
Swissmedic Publishes swissdamed Machine-to-Machine REST API Documentation (Version 1.0)
Swissmedic publishes Version 1.0 of the swissdamed Machine-to-Machine REST API documentation for UDI registration and market status management.
MHRA Updates Guidance on Regulation of Medical Devices in Northern Ireland
MHRA updates guidance on medical device regulation in Northern Ireland. Key impacts for manufacturers under EU MDR and IVDR.
MHRA Updates Guidance on Regulating Medical Devices in the UK
MHRA updates guidance on regulating medical devices in Great Britain and Northern Ireland. Key rules on UKCA, CE marking and registration.
MHRA Updates Guidance on Registration of Medical Devices for the UK Market
MHRA updates UK medical device registration rules, introduces annual Level 2 GMDN-based fees from April 2026 and confirms NI EUDAMED transition.
FDA Updates Recognized Biocompatibility Standards – Recognition List No. 065
FDA Recognition List 065 updates biocompatibility standards, including ISO 10993-4 Amendment 1 and multiple ASTM revisions. Impact for manufacturers.
Team-NB Publishes New Position Paper on Safety and Performance of Reagent Devices Used in Combination
New Team-NB position paper clarifies IVDR expectations for safety, risk assessment and performance evaluation of reagent devices used in combination.
European Commission Publishes Updated Harmonised Standards Under MDR
The European Commission adopts Implementing Decision (EU) 2026/193, updating harmonised standards under MDR (EU) 2017/745.
MHRA Publishes Updated Guide on Defective Medicinal Products
The MHRA has published its February 2026 Guide on Defective Medicinal Products. Learn what UK manufacturers and marketing authorisation holders must do regarding defect reporting, recalls, and DMRC obligations.
ANVISA Publishes Draft Normative Instruction on UDI Data Transmission and SIUD Management
ANVISA publishes draft rules on UDI data transmission and SIUD management in Brazil. Learn how the new requirements impact medical device manufacturers.
FDA Publishes Draft ICH E22 Guideline on Patient Preference Studies for Public Consultation
The FDA has published a draft guidance adopting the ICH E22 guideline on Patient Preference Studies. Learn what this means for manufacturers involved in global development programmes.
Swissmedic Publishes Updated Online Help for the Medical Device Publication Service
Swissmedic has published version 1.2 of its guidance on the Medical Device Publication Service, clarifying how Field Safety Corrective Actions and Field Safety Notices are published and accessed.
Swissmedic Updates Formal Requirements Guidance – Version 19.0 Applicable from February 2026
Swissmedic has published version 19.0 of its Formal Requirements Guidance, applicable from February 2026. Learn what manufacturers and combination product developers need to prepare for.
FDA Issues Updated Guidance on Computer Software Assurance for Production and QMS Software
The FDA has issued an updated guidance on Computer Software Assurance for production and quality management system software, aligned with the new QMSR and ISO 13485.