Swissmedic Updates swissdamed UDI Data Dictionary
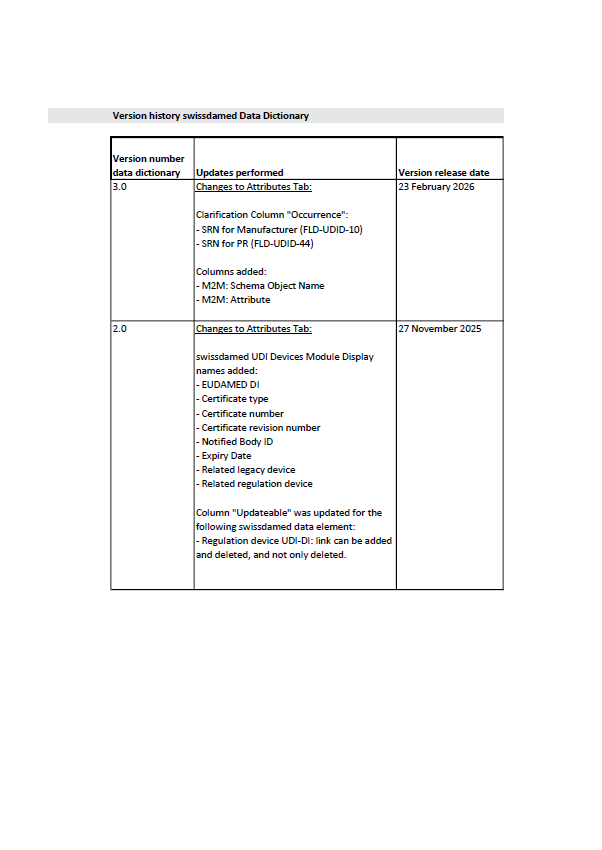
Swissmedic has released Version 3.0 of the swissdamed UDI Data Dictionary, introducing technical clarifications and updates to the data structure used for device information in the Swiss database.
According to the version history, the latest update introduces clarifications to the “Occurrence” column for two fields:
SRN for Manufacturer (FLD-UDID-10)
SRN for PR (FLD-UDID-44)
In addition, two machine-to-machine (M2M) columns were added to the attributes tab:
M2M: Schema Object Name
M2M: Attribute
These additions appear intended to improve technical mapping and system integration for data exchanges with the swissdamed database.
The document also recalls updates introduced in Version 2.0 (November 2025), including additional fields displayed in the swissdamed UDI Devices module, such as EUDAMED DI, certificate information, Notified Body ID, expiry date, and links to legacy or regulation devices.
The swissdamed data dictionary continues to reference several EUDAMED technical documentation elements, including data dictionaries, enumerations, and XML schemas used in the EU database framework.
Manufacturers placing devices on the Swiss market should review the updated dictionary to ensure alignment of their UDI data structures and system integrations with the latest swissdamed specifications.
Read the full document below.