FDA Updates Recognized Biocompatibility Standards – Recognition List No. 065
FDA Recognition List 065 updates biocompatibility standards, including ISO 10993-4 Amendment 1 and multiple ASTM revisions. Impact for manufacturers.
Team-NB Publishes New Position Paper on Safety and Performance of Reagent Devices Used in Combination
New Team-NB position paper clarifies IVDR expectations for safety, risk assessment and performance evaluation of reagent devices used in combination.
European Commission Publishes Updated Harmonised Standards Under MDR
The European Commission adopts Implementing Decision (EU) 2026/193, updating harmonised standards under MDR (EU) 2017/745.
MHRA Publishes Updated Guide on Defective Medicinal Products
The MHRA has published its February 2026 Guide on Defective Medicinal Products. Learn what UK manufacturers and marketing authorisation holders must do regarding defect reporting, recalls, and DMRC obligations.
ANVISA Publishes Draft Normative Instruction on UDI Data Transmission and SIUD Management
ANVISA publishes draft rules on UDI data transmission and SIUD management in Brazil. Learn how the new requirements impact medical device manufacturers.
FDA Publishes Draft ICH E22 Guideline on Patient Preference Studies for Public Consultation
The FDA has published a draft guidance adopting the ICH E22 guideline on Patient Preference Studies. Learn what this means for manufacturers involved in global development programmes.
Swissmedic Publishes Updated Online Help for the Medical Device Publication Service
Swissmedic has published version 1.2 of its guidance on the Medical Device Publication Service, clarifying how Field Safety Corrective Actions and Field Safety Notices are published and accessed.
Swissmedic Updates Formal Requirements Guidance – Version 19.0 Applicable from February 2026
Swissmedic has published version 19.0 of its Formal Requirements Guidance, applicable from February 2026. Learn what manufacturers and combination product developers need to prepare for.
FDA Issues Updated Guidance on Computer Software Assurance for Production and QMS Software
The FDA has issued an updated guidance on Computer Software Assurance for production and quality management system software, aligned with the new QMSR and ISO 13485.
EUDAMED Production v2.22.0: What Manufacturers Need to Know
EUDAMED Production v2.22.0 introduces EMDN versioning, improved traceability, and DTX updates. Learn what this means for manufacturers.
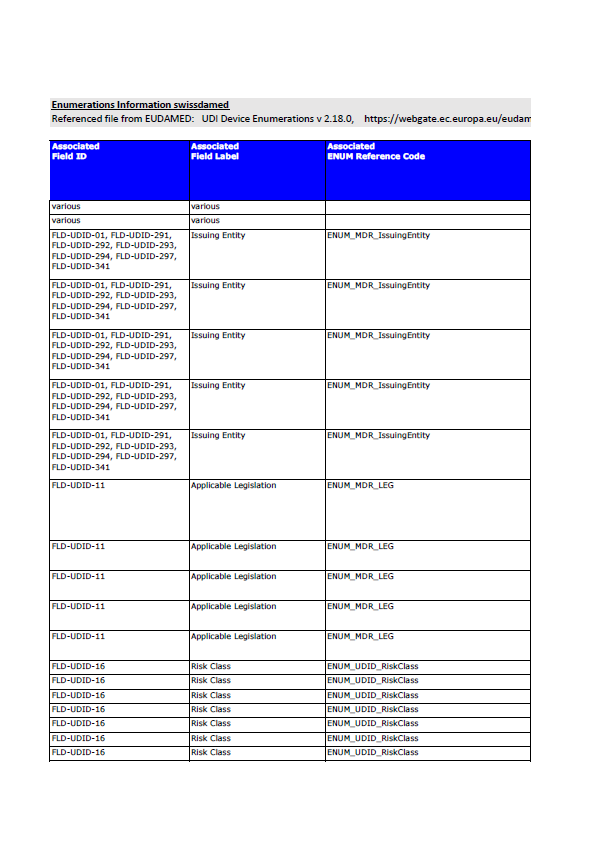
Swissmedic Publishes Updated EUDAMED UDI Enumerations: What Manufacturers Should Know
Swissmedic publishes updated EUDAMED UDI enumerations (v2.18.0). What this means for manufacturers preparing UDI, device and certificate data submissions.
Swissmedic Updates eIAM Portal Administrator Guidance – Version 3.4 Effective February 2026
Swissmedic publishes eIAM Portal Administrator guidance version 3.4, effective February 2026. Learn what this means for manufacturers using Swiss eGov services.
FDA Issues Updated Final Guidance on Cybersecurity in Medical Devices
FDA issues final guidance on cybersecurity in medical devices, outlining QMS, premarket documentation, and Section 524B requirements for manufacturers.
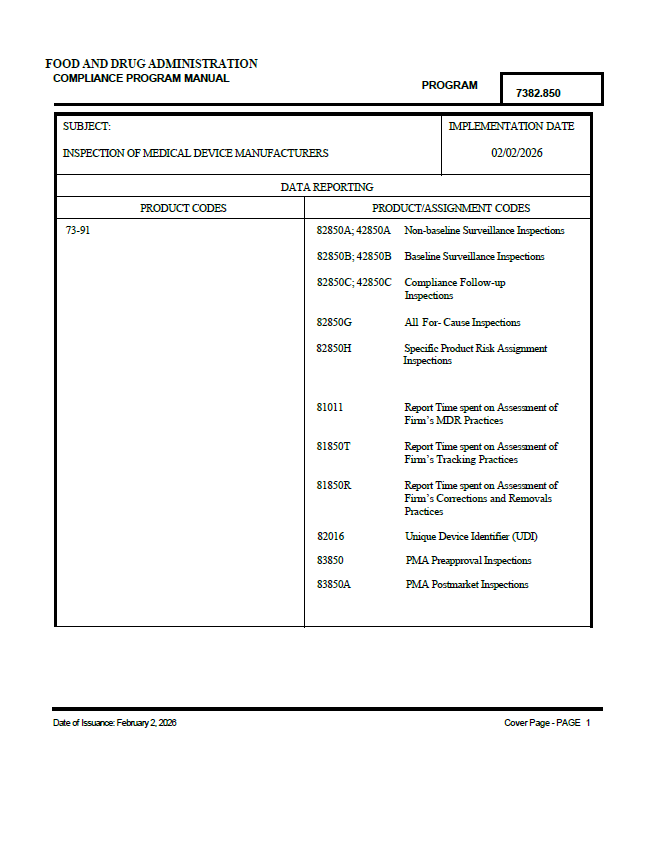
FDA Implements Updated Medical Device Inspection Compliance Program
FDA implements Compliance Program 7382.850, aligning medical device inspections with the Quality Management System Regulation and ISO 13485.
European Commission Publishes New Harmonised Standards for Sterilisation and IVD Labelling under IVDR
The European Commission has adopted Commission Implementing Decision (EU) 2026/197 of 28 January 2026, amending Implementing Decision (EU) 2021/1195 by adding new references to harmonised standards supporting Regulation (EU) 2017/746 on in vitro diagnostic medical devices (IVDR). The Decision was published in the Official Journal of the European Union on 30 January 2026 and entered into force on the same day.
EU-U.S. Data Privacy Framework Updated: What It Means for Medical Device Manufacturers
New EU-U.S. Data Privacy Framework FAQ impacts data transfers to U.S. vendors. What this means for medical device manufacturers in 2026.
MedTech Europe Proposes Targeted Reforms to MDR and IVDR – Strategic Input for 2026 Regulatory Revisions
MedTech Europe’s MDR/IVDR reform proposals remain key as the EU prepares its 2026 legislative update. Find out what this means for manufacturers.
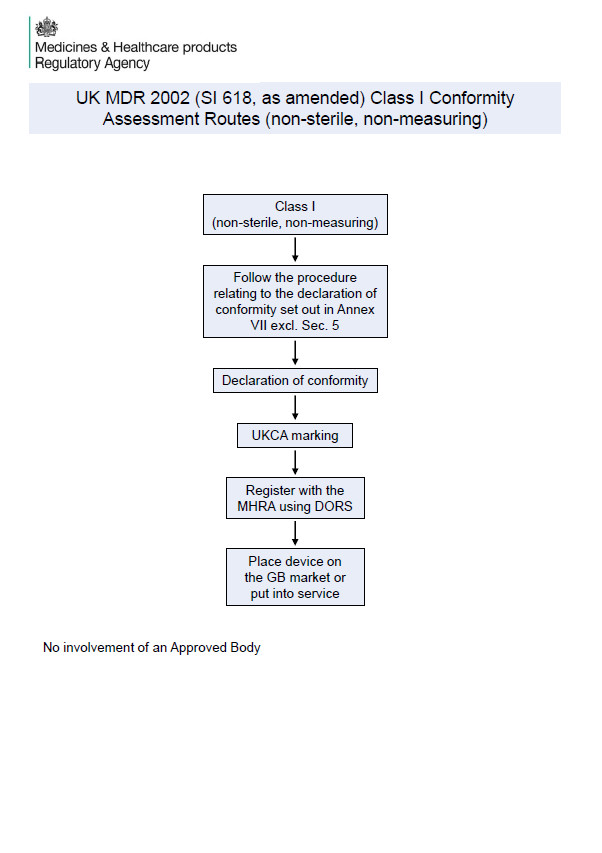
New MHRA Summary: UK Conformity Assessment Routes for All Device Classes
New MHRA guidance summarises UK conformity routes for all medical device classes. Essential for UKCA compliance and UK MDR planning.
FDA Publishes Updated Guidance on Clinical Decision Support Software
The FDA has issued updated guidance on Clinical Decision Support (CDS) software, clarifying which software functions are excluded from the medical device definition under U.S. law.
Australia Updates Guidance on Export Certificates for Medical Devices: What EU Manufacturers Need to Know
The TGA has updated its guidance on Certificates of Free Sale and Export Certificates for medical devices. Key changes for EU manufacturers exporting via Australia.