Swissmedic Updates swissdamed Actors User Guide: What Manufacturers and Authorised Representatives Should Know
Swissmedic has updated the swissdamed Actors User Guide (v2.0), introducing AGOV login migration updates, mandate transfer functionality, and clarified actor management requirements for medical device manufacturers and authorised representatives in Switzerland.
European Commission Updates MIR 7.3.1 Vigilance Reporting Forms
The European Commission has updated the MIR 7.3.1 vigilance reporting forms under the EU MDR and IVDR. Manufacturers should verify accepted versions and updated XSD/XSL files.
FDA Launches One-Day Inspectional Assessments Pilot
The FDA has launched a pilot program introducing one-day inspectional assessments across medical products, biologics, clinical research, and food sectors to support more targeted and efficient oversight.
TEAM-NB Updates Code of Conduct for Notified Bodies (Version 5.2 – April 2026)
TEAM-NB releases Code of Conduct v5.2 for MDR/IVDR Notified Bodies. Discover key changes and how they impact medical device manufacturers.
FDA Issues Draft Guidance on Compliance Policy for Certain NIOSH-Approved Respirators
FDA releases draft guidance proposing reduced regulatory requirements for certain NIOSH-approved respirators. Understand the impact on medical device manufacturers.
Swissmedic Publishes Updated Information Sheet on Medical Device Software (Version 3.0)
Swissmedic releases updated guidance on medical device software (MDSW), covering qualification, classification, and regulatory requirements under MDR/IVDR.
EU Batteries Regulation (EU) 2023/1542: Ongoing Implementation and Considerations for Manufacturers
Learn about EU Batteries Regulation (EU) 2023/1542, including key requirements, scope, and timeline. What manufacturers need to know for compliance from 2025 onwards.
EU Defines Conditions for Delaying Cybersecurity Notifications under the Cyber Resilience Act
EU Delegated Regulation 2026/881 defines when cybersecurity vulnerability and incident notifications may be delayed under the Cyber Resilience Act.
MDCG Publishes Updated Guidance on Medical Device Classification (MDCG 2021-24 Rev.1)
MDCG updates medical device classification guidance under EU MDR. Key changes manufacturers must review for compliance and documentation.
EDPB Releases New 2026 DPIA Template for Public Consultation
The EDPB publishes a new 2026 DPIA template to standardise GDPR compliance. What it means for medical device manufacturers.
Notified Bodies Raise Concerns on Targeted MDR/IVDR Revision
Notified bodies highlight key risks and proposals in the EU MDR/IVDR targeted revision, including clinical evidence, WET, and regulatory oversight.
European Parliament Adopts Amendments to Simplify AI Act Implementation (“Digital Omnibus on AI”)
EU Parliament adopts AI Act simplification package. Key changes impact high-risk AI timelines, MDR alignment, and compliance requirements.
Swissmedic publishes updated guidance on Systems and Procedure Packs (SPP)
Swissmedic updates guidance on systems and procedure packs. Key obligations, CH-REP requirements, and impact for medical device manufacturers.
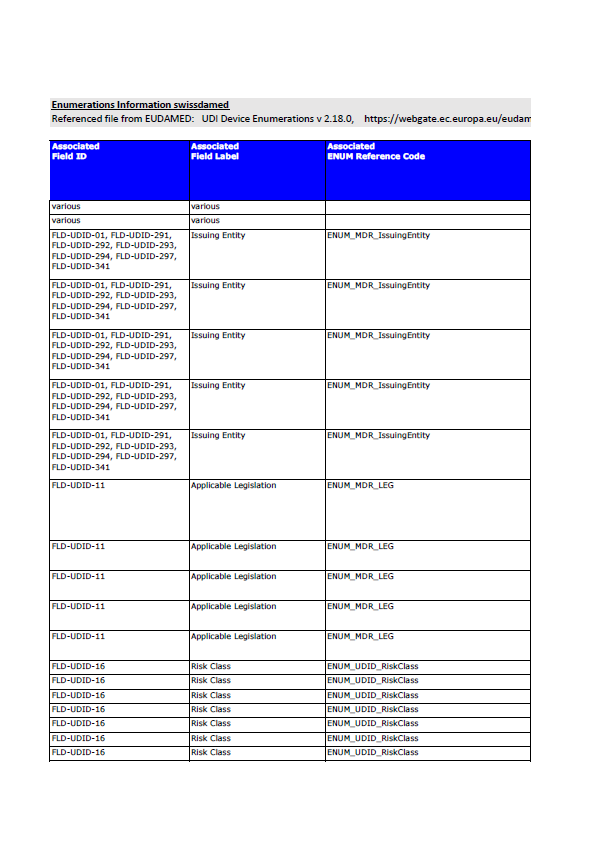
Swissmedic Publishes Updated EUDAMED UDI Enumerations: What Manufacturers Should Know
Swissmedic publishes updated EUDAMED UDI enumerations (v2.18.0). What this means for manufacturers preparing UDI, device and certificate data submissions.
EU-U.S. Data Privacy Framework Updated: What It Means for Medical Device Manufacturers
New EU-U.S. Data Privacy Framework FAQ impacts data transfers to U.S. vendors. What this means for medical device manufacturers in 2026.
FDA Publishes Final Guidance on Computer Software Assurance: Key Implications for Manufacturers
The FDA has released its final guidance on Computer Software Assurance (CSA) for production and quality system software, introducing a risk-based, streamlined approach to validating software used in manufacturing and quality operations. The updated framework helps manufacturers reduce unnecessary documentation, focus validation efforts on functions that impact product quality and patient safety, and accelerate the adoption of automated and digital technologies while remaining fully compliant with FDA requirements.
FDA Issues Updated Scientific Recommendations for Biosimilars: What Manufacturers Need to Know
The FDA has issued a new draft guidance that updates the scientific principles for demonstrating biosimilarity. The document clarifies when strong analytical similarity data, supported by comparative human pharmacokinetic and immunogenicity assessments, may be sufficient to demonstrate biosimilarity without requiring a comparative clinical efficacy study. This updated approach can help manufacturers reduce development timelines and costs, while placing greater emphasis on robust analytical packages, sensitive PK study designs and a clear, risk-based justification aligned with FDA expectations.
MDCG 2019-11 Rev.1 - New guidance on qualification and classification of software as a medical device or in vitro diagnostic medical device
The new revised version of the MDCG 2019-11 (Rev.1) clarifies the qualification and classification of software as a medical device (MDSW ) and in vitro diagnostic software (IVD), in the context of the MDR and IVDR. The document covers classification rules, integration with AI Act and EHDS, and includes practical and up-to-date examples. Smart MDR supports MDSW manufacturers in managing compliance and technical documentation.
UK publishes new model periodic safety report (PSUR) for medical devices - MHRA 2024
The MHRA has published a new Periodic Safety Update Report (PSUR) template for medical devices in the UK, in line with the UK MDR. The document details how manufacturers should structure their post-market monitoring (PMS) reports, including performance data, surveillance, PMCF and benefit-risk profile analysis.
Health Canada publishes new guidelines on the use of technical standards to demonstrate compliance with the Medical Devices Regulations
In January 2025, Health Canada published a new guide on the use of recognized standards to demonstrate compliance with the requirements of the Medical Devices Regulations. The document guides manufacturers in preparing submissions, updates and lifecycle management of applicable standards.