MHRA Updates Guidance on Regulating Medical Devices in the UK
MHRA updates guidance on regulating medical devices in Great Britain and Northern Ireland. Key rules on UKCA, CE marking and registration.
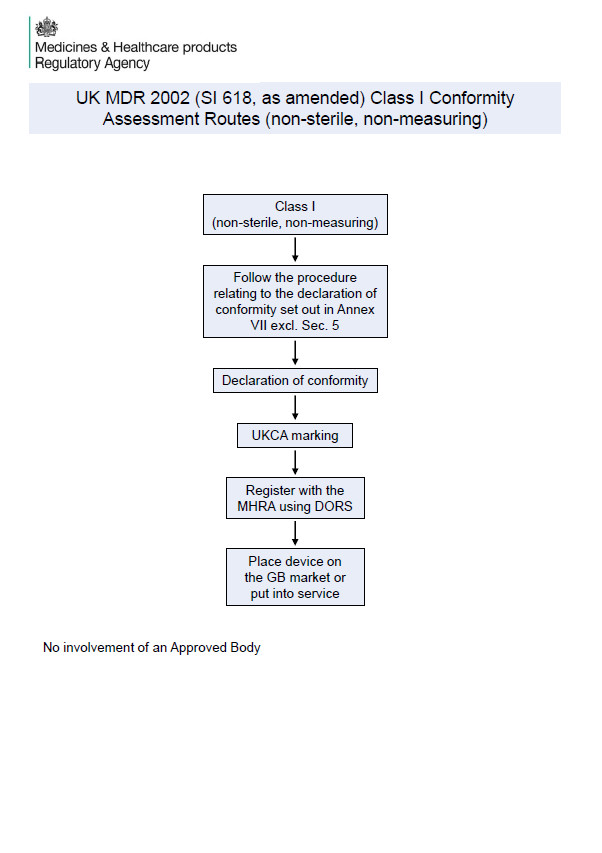
New MHRA Summary: UK Conformity Assessment Routes for All Device Classes
New MHRA guidance summarises UK conformity routes for all medical device classes. Essential for UKCA compliance and UK MDR planning.
MHRA Publishes Updated Guidance on Clinical Investigations for Medical Devices: Key Implications for Manufacturers
The MHRA has published updated guidance on clinical investigations for medical devices, detailing the requirements manufacturers must meet when planning and conducting investigations in Great Britain. The update strengthens documentation expectations, safety requirements and regulatory oversight, making it essential for all manufacturers preparing UK submissions.