Health Canada Mandates the Use of REP for Medical Device Submissions
Health Canada now requires mandatory use of the Regulatory Enrolment Process (REP) and CESG for Class II–IV medical device submissions. Learn what manufacturers must do and how the new IMDRF ToC requirements may impact regulatory workflows.
Swissmedic Publishes “Good Practice for Materiovigilance in Hospitals” (GPMV-Spital)
Swissmedic releases GPMV-Spital, a new framework for materiovigilance in hospitals. Understand the impact on medical device manufacturers and post-market surveillance.
FDA Issues Final Guidance on Medical Devices with Indications Associated with Weight Loss
The FDA has issued final guidance on medical devices with indications associated with weight loss, outlining regulatory expectations for clinical studies, non-clinical testing, and benefit-risk evaluation.
MHRA Updates Guidance on Registration of Medical Devices for the UK Market
MHRA updates UK medical device registration rules, introduces annual Level 2 GMDN-based fees from April 2026 and confirms NI EUDAMED transition.
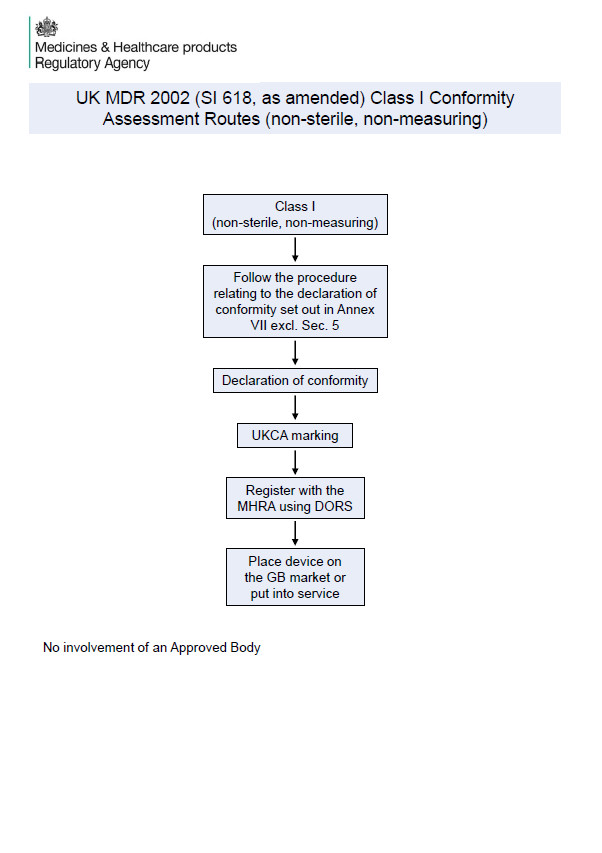
New MHRA Summary: UK Conformity Assessment Routes for All Device Classes
New MHRA guidance summarises UK conformity routes for all medical device classes. Essential for UKCA compliance and UK MDR planning.
Australia Updates Guidance on Export Certificates for Medical Devices: What EU Manufacturers Need to Know
The TGA has updated its guidance on Certificates of Free Sale and Export Certificates for medical devices. Key changes for EU manufacturers exporting via Australia.